 |
 |
- Search
| J Korean Med Assoc > Volume 66(4); 2023 > Article |
|
Abstract
Background: Patients with chronic myeloid leukemia (CML) now have an improved life expectancy similar to that of the general population due to the introduction of tyrosine kinase inhibitors (TKIs). However, many patients experience mild to severe adverse events while undergoing TKI treatment. This review aimed to discuss the adverse events of TKIs, including myocardial infarction and hypertension, and comprehensively analyze strategies for minimizing vascular complications.
Current Concepts: Near-fatal cardiovascular events (CVEs) are common among patients receiving nilotinib and ponatinib. However, those receiving other TKIs, such as imatinib and dasatinib, rarely experience CVEs. Among these CVEs, vascular complications, including peripheral arterial occlusion, venous occlusion, and hypertension, are exacerbated in patients with pre-existing vascular risk factors and prolonged TKI use. Therefore, it is crucial to assess predisposing factors to vascular complications and select the optimal TKI to minimize serious CVEs before initiating therapy. Additionally, patients should be closely monitored for vascular complications during nilotinib and ponatinib treatment.
Discussion and Conclusion: Despite advancements in therapeutic approaches and research on CML leading to the development of target-specific TKIs aiming to minimize side effects, newer generations are not entirely devoid of adverse events. Hence, it is important for patients and physicians to be knowledgeable about these medications to effectively monitor for side effects, particularly those that are life-threatening, such as vascular toxicity. It is now more important than ever to carefully observe symptoms and perform adequate testing to identify at-risk individuals early and avoid preventable adverse events.
Current Concepts: Near-fatal cardiovascular events (CVEs) are common among patients receiving nilotinib and ponatinib. However, those receiving other TKIs, such as imatinib and dasatinib, rarely experience CVEs. Among these CVEs, vascular complications, including peripheral arterial occlusion, venous occlusion, and hypertension, are exacerbated in patients with pre-existing vascular risk factors and prolonged TKI use. Therefore, it is crucial to assess predisposing factors to vascular complications and select the optimal TKI to minimize serious CVEs before initiating therapy. Additionally, patients should be closely monitored for vascular complications during nilotinib and ponatinib treatment.
Discussion and Conclusion: Despite advancements in therapeutic approaches and research on CML leading to the development of target-specific TKIs aiming to minimize side effects, newer generations are not entirely devoid of adverse events. Hence, it is important for patients and physicians to be knowledgeable about these medications to effectively monitor for side effects, particularly those that are life-threatening, such as vascular toxicity. It is now more important than ever to carefully observe symptoms and perform adequate testing to identify at-risk individuals early and avoid preventable adverse events.
만성골수성백혈병(chronic myeloid leukemia, CML)은 필라델피아(Philadelphia, Ph) 염색체 이상의 결과로 발현된 BCR-ABL1 타이로신인산화효소(tyrosine kinase)가 골수구(myeloid) 계열 세포를 과도하게 증식시키고 세포 사멸을 억제함으로 발병하는 혈액암이다[1]. 과거 CML은 하이드록시유레아(hydroxyurea)나 인터페론(interferon), 또는 공여자가 있는 경우 조혈모세포이식(allogenic stem cell transplantation)이 주된 치료였으나, 2000년대 표적치료제인 타이로신인산화효소 억제제(tyrosine kinase inhibitor, TKI)의 등장으로 CML 치료의 패러다임은 완전히 바뀌게 되었다[2]. TKI는 BCR-ABL1의 아데노신 삼인산(adenosine triphosphate, ATP) 부착 부위에 대신 결합하여 인산화효소의 활성화를 막아 병의 진행을 억제하는데, 1세대 TKI인 이마티닙(imatinib)은 매일 한번의 경구 약물 복용만으로 CML 치료에 월등하게 우수한 효과를 보여주었다[3]. 뒤 이은 2세대 TKI인 다사티닙(dasatinib)과 닐로티닙(nilotinib)은 이마티닙 대비 더 빠르고 더 깊은 수준의 치료 반응을 보여주었으며, T315I와 같은 TKI 불응 또는 내성 돌연변이를 가지는 환자에서도 효과를 보이는 3세대 TKI인 포나티닙(ponatinib)과 최근 4세대 TKI인 애시미닙(asciminib)이 개발됨에 따라 CML은 더는 불치의 병이 아닌 조절이 가능한 만성 질환에 근접하게 되었다[4]. 그러나 TKI 도입 20여 년이 지난 지금 다른 한편에서는 오랜 기간 TKI를 복용함으로 누적되는 문제들이 CML 치료의 장애로 떠오르고 있다. 평생 치료에 따른 의료 비용의 누적과 TKI 관련 독성으로 인한 합병증, 그리고 환자 삶의 질 하락 등으로 요약할 수 있다. 특히 치료 효과가 강력하다고 알려진 2세대, 3세대 TKI의 경우 뇌졸중이나 심근경색, 말초 혈관 폐쇄와 같은 혈관 관련 합병증이 빈번하고 이로 인해 환자의 생명이 위협받는 경우들이 나타나면서, 치료 중 합병증 관리가 어느 때보다 중요해지고 있다. 이에 이 논문에서는 CML 환자에서 TKI 사용 중 발생할 수 있는 심혈관계 부작용 및 고혈압 등의 혈관 관련 합병증에 대해 고찰하였다.
32세 남자 환자가 2012년 10월 몸무게 감소 및 피곤함, 그리고 복부 팽만감으로 외래 내원하였다. 혈액검사상 백혈구 509,000/µL, 혈색소 13.6 g/dL, 혈소판 154,000/µL로 골수검사(bone marrow biopsy)를 시행하여 CML-만성기(chronic phase, CP)로 확인되었으며, 중합효소사슬반응(polymerase chain reaction, PCR)을 통해 BCR-ABL1 유전자가 확인되어 최종적으로 CML-CP로 진단되었다. Sokal 점수 및 Hasford 점수에서 각각 중증도였으며, 고혈압이나 당뇨와 같은 기저 질환 없는 상태로 닐로티닙 300 mg 하루 2회 복용 치료를 시작하였다. 치료 시작 후 1년 검사에서 BCR-ABLinternational scale (IS) 값은 0.971%로 주요 분자학적 반응(major molecular response, MMR)의 획득은 실패하였으나, 말초 혈액검사상 완전 혈액학적 반응(complete hematologic response, CHR) 및 골수검사에서 Ph 염색체가 확인되지 않은 완전 세포학적 반응(complete cytogenetic response, CCyR)을 이루어 닐로티닙 치료를 유지하였다. 치료 후 2년 3개월이 지난 2015년 1월, 급성 흉통으로 로컬 병원 응급실에 내원하여 심실 세동(ventricular fibrillation)으로 응급심폐소생술을 2차례 시행하고, 심전도상 ST 분절 상승이 확인되어 ST 분절 상승 급성심근경색(ST elevation myocardial infarction)으로 응급 관상동맥조형술(coronary arteriography, CAG)을 시행하였다. CAG에서 근위부 전하동맥(proximal anterior descending coronary artery)의 완전폐쇄(total occlusion)가 확인되어 스텐트를 삽입하였다. 이후 이마티닙 400 mg 하루 1회 복용으로 변경하고, 여전히 MMR 획득은 실패하였으나, BCR-ABLIS 유지로 치료를 지속하였다. 2019년 9월 BCR-ABLIS 값이 31.5434%로 상승하여 ABL1 인산화효소의 대표 8개 도메인에 대해 내성검사를 시행하였으나 내성 돌연변이가 확인되지 않아 이마티닙 600 mg으로 증량하였다. 이후 BCR-ABLIS 값이 점차 감소하여, 2022년 8월 BCR-ABLIS 0.2059%로 감소되어 이마티닙 40 0mg으로 감량하고 CHR 상태 유지 중이며, 추가적인 심혈관 질환은 현재까지 발생하지 않고 있다.
닐로티닙의 3상 연구인 Evaluating Nilotinib and Safety in Clinical Trials-Newly Diagnosed Patients (ENESTnd)에서는 12개월 MMR 획득률에서 44% 대 22% (P<0.001)로, 닐로티닙 300 mg 1일 2회 복용하는 군이 이마티닙 400 mg 1일 1회 복용하는 군에 비해 우월한 치료 성적과 비교적 안정적인 부작용 발생을 보여주었다[5]. 그러나 여러 후향 연구들에서 닐로티닙을 복용하는 환자들이 말초 동맥혈관 폐쇄 질환(peripheral artery occlusive disease, PAOD)의 발생이 높고, 일부의 경우 하지를 절단하는 경우까지 있는 것으로 보고하였다[6,7]. ENESTnd 연구 추적 결과에서도 닐로티닙 투약군에서 PAOD 뿐만 아니라 심근경색, 뇌졸중과 같은 심혈관 질환(cardiovascular events, CVE)이 이마티닙 투약군 대비하여 부작용 등급에 상관없이 7.5% 대 2.1%의 높은 비율로 보고되었다[8]. 이러한 CVE 발생의 위험성은 닐로티닙의 용량이 높을수록, 투약 기간이 길수록, 그리고 환자의 나이, 성별, 흡연력, 콜레스테롤 수치, 혈압 등을 고려하여 결정되는 관상동맥질환 위험도(Framingham risk score) [9]가 높을수록 증가하는 경향을 보였다. 최근에 발표된 ENESTnd의 10년 최종 추적 결과에 따르면 Framingham risk score가 20% 이상이며, 닐로티닙을 400 mg 1일 2회, 5년 이상 복용한 경우 CVE 발생은 33.3%까지 보고된 반면, Framingham risk score가 10% 미만이고, 닐로티닙을 300 mg 1일 2회, 5년 이내로 복용하는 경우는 2.2%의 CVE 발생이 보고되었다. 그에 비해 이마티닙을 복용한 환자들은 Framingham risk score가 높고 5년 이상 복용을 하였더라도 4.8%만이 CVE를 경험하였다(Table 1) [10]. 국내 단일 센터 연구결과에서도 비슷한 경향이 확인되었는데, 혈압, 당뇨, 고지혈증, 흡연력, 그리고 이전 심혈관 질환 중 1가지 이상의 혈관 관련 위험인자를 가진 경우, 누적 CVE의 발생은 닐로티팁 5년 복용 시 14.5%에서 10년 이상 복용할 시 43.6%으로 증가하는 것으로 확인되었다. 반면, 혈관 관련 위험인자가 없는 경우는 10년 이상 복용 시 14.5%의 누적 CVE를 보여주었다.
닐로티닙의 혈관 관련 독성에 대한 기전은 명확히 밝혀지지는 않았으나 여러가지 다양한 요인들이 복합적으로 영향을 끼쳤을 것으로 생각된다(Figure 1). 닐로티닙은 혈관 내피세포에 동맥경화와 관련한 단백질들을 많이 생성하도록 조절하는 한편, 동맥경화를 개선할 수 있는 혈관 내피세포의 증식 및 이동을 억제함으로 동맥경화를 악화시킬 수 있는 것으로 알려졌다[11]. 또한 닐로티닙이 ABL1 인산화 효소에 결합하는 것 외에도 혈관 복구 및 동맥경화와 관련 있는 인산화효소에도 결합을 함으로 혈관 독성을 유발하였을 가능성도 제시되었다[11]. 닐로티닙 투약된 사람과 쥐의 혈액에서는 동맥 혈류가 흐르는 가운데에서도 혈전 형성이 증가되는 것이 관찰되었는데, 닐로티닙이 혈관 내피세포 및 혈소판 활성화와 관련된 단백질 생성을 증가시켜 혈관 내 혈소판 부착을 촉진하고, 염증세포와 사이토카인 생성을 촉진함으로 결과적으로 혈전 형성에 기여할 것으로 생각된다[12]. 한편 ENESTnd 연구에서 닐로티닙의 투약은 CVE 발생의 증가 외에도 이마티닙 대비하여 혈액 내 콜레스테롤(27.6% 대 3.9%) 및 혈당(7.2% 대 0.4%) 수치 상승도 함께 보고한 바 있다[8]. 실제 여러 후향 연구에서도 대사 관련한 유사한 결과가 제시되었는데, 대사 장애(metabolic dysfunction)가 심혈관 질환의 위험인자임을 감안한다면, 닐로티닙을 복용하는 환자에서 높게 보고되는 CVE는 콜레스테롤 및 혈당 상승이 원인이 되었을 가능성도 배제할 수 없다[13]. 이와는 반대로 이마티닙은 오히려 혈관 건강에 긍정적인 영향을 끼친다는 것이 보고되었는데, 이마티닙은 혈액 내 아디포넥틴(adiponectin) 수치를 높임으로 혈당을 낮출 뿐만 아니라, 또 다른 인산화효소인 혈소판 유래 성장인 자수용체(platelet derived growth factor receptor)를 억제함으로 당뇨와 관련 있는 동맥경화를 억제하는 결과를 보 여주었다[14,15]. 닐로티닙과 이마티닙의 3상 연구를 모아 PAOD의 위험도를 확인한 메타분석 연구에 따르면 닐로티닙을 복용했던 환자들의 PAOD 발생률은 1.3%였던 반면, 이마티닙을 복용한 환자들은 0.2%, TKI를 복용하지 않은 환자들의 PAOD는 0.6%였다[16]. 약물 투약시간을 보정하지 않은 단순 PAOD 발생률로 비록 해석에 주의가 필요하나, 닐로티닙과 이마티닙의 혈관 관련 독성의 위험도 차이에 대해 시사하는 바가 있다.
3세대 TKI인 포나티닙은 2상 연구인 Ponatinib Ph+ ALL and CML Evaluation (PACE) 임상시험에서 연구 시작 24개월만에 11.8%의 심각한 동맥 혈전 부작용이 보고되어 일시적으로 환자 등록이 중단된 바 있다[17]. 그러나 T315I를 비롯하여 돌연변이 발생으로 이전 TKI 치료에 실패한 CML 환자들에게 다른 대안이 없음이 고려되어 임상은 곧 재개되었고, PACE 5년 최종 추적 결과에서 31%의 CVE와 6%의 정맥 혈전 발생이 보고되었다[18]. CVE 발생은 포나티닙 용량 의존적이어서 치료 반응에 따라 적극적으로 용량을 조절하는 임상이 진행되었는데, Optimizing Ponatinib Treatment in CP-CML (OPTIC) 임상시험에서는 총 282명의 환자가 1:1:1로 나누어 45 mg, 30 mg, 15 mg으로 치료를 시작하였다. OPTIC 연구에서는 BCR-ABLIS 값과 CVE의 발생을 관찰하며 적극적으로 용량을 조절하였는데, 그 결과 12개월째 ABL IS ≤1% 기준으로 본 치료 효과는 44.1%, 29.0%, 23.1%로 처음 시작 시 45 mg으로 시작한 군이 가장 높았으나, 그에 비해 CVE의 발생은 5명, 5명, 3명으로 큰 차이를 보이지 않았다(Table 2) [19]. 따라서 포나티닙은 혈관 관련 위험인자와 돌연변이 정도를 고려하여 45 mg, 30 mg, 15 mg 용량 중 선택하여 투여하되 치료 반응과 부작용을 보며 용량을 조절하는 것이 권고된다[20]. 포나티닙의 혈관 관련 독성과 관련한 기전은 명확히 알려지지는 않았으나, 닐로티닙이 보여주었던 혈관 독성 기전과는 다소 차이가 있을 것으로 생각된다. 이는 포나티닙이 닐로티닙과는 달리 오히려 혈소판의 활성화와 응집을 억제하고[21], 혈관 내피세포 성장인자수용체(vascular endothelial growth factor receptor, VEGFR)에 결합하여 강력한 억제 효과를 보이기 때문이다[13]. 이전 연구들에서 VEGF 억제를 하는 TKI들이 고혈압이나 혈전 색전증, 심혈관 질환의 위험성이 높였던 것을 떠올려 본다면, 포나티닙의 오프 타켓(off-target) 효과가 CVE의 발생에 영향을 끼쳤을 가능성도 추측해 볼 수 있다[22]. 한편 포나티닙도 이전 심혈관 질환에 대한 위험인자를 가지고 있는 환자들에게 높게 보고되는 경향이 있다[13,18,19].
이전 TKI 와는 다른 생물학적 메커니즘으로 작동하는 애시미닙은 포나티닙과 함께 이전 치료에 불응을 보이거나 T315I 돌연변이가 있는 CML환자에서도 효과적인 4세대 TKI이다[23]. 애시미닙은 Specifically Targets the ABL Myristoyl Pocket (STAMP) 3상 연구에서 이전 2개 이상의 TKI 치료에 실패한 환자들이 포함된 연구였음에도 불구하고 우수한 치료 효과와 보수티닙(bosutinib) 대비하여 중증 부작용은 50.6% 대 60.5%, 부작용에 의한 치료 중단 비율은 5.8% 대 21.1%로 우수한 부작용 분포를 보여주었다[24]. 따라서 T315I 돌연변이가 있고 CVE 발생 가능성이 높은 경우라면, 포나티닙보다는 애시미닙 선택을 고려해 볼 수 있다. 그러나 애시미닙으로 치료한 환자에서도 CVE 발생이 일부 보고되어 여전히 주의는 필요하다. STAMP 연구의 최근 결과에서2.3년의 추적관찰 기간 동안 보수티닙 복용한 환자 중 1명(1.3%)만이 CVE를 겪은 반면, 애시미닙 환자군에서는 총 8명(5.1%)의 환자들이 CVE를 겪었기 때문이다(Table 2) [25]. 다만 이들 환자 대부분이 이전에 포나티닙 또는 닐로티닙 등 여러 TKI로 치료를 받았고 8명 중 6명은 CVE 관련 위험인자를 가지고 있었던 것으로 조사되어 애시미닙의 정확한 CVE 발생 위험성에 대해서는 추가적인 연구가 필요하겠다.
55세 여자가 2022년 4월 어지러운 증상으로 로컬 병원에서 혈액검사를 시행하고, 백혈구 상승으로 내원하였다. 혈액검사상 백혈구 456,200/µL, 혈색소 10.1 g/dL, 혈소판 388,000/µL으로 PCR 검사에서 BCR-ABL1 유전자가 확인되어 최종적으로 CML-CP로 진단되었다. 비장 크기는 3 cm였으며, Sokal 점수 및 Hasford 점수에서 각각 중증도로 확인되었고, 닐로티닙 300 mg 하루 2회 복용을 시작하였다. TKI 복용 중 3단계의 혈소판 감소가 확인되어 약물 복용을 일시 중단하였고, 치료 재개 이후 다시 피부 발진으로 200 mg 하루 2회로 감량하였다. 2022년 7월 말 혈액검사에서 미성숙 백혈구 20%가 확인되어 골수검사 시행하고, CML-급성기(blast phase, BP)로 진행하였음을 확인하였다. 돌연변이 검사에서는 닐로티닙에 불응을 보이는 Y253H가 확인되었다. 환자는 원래 알고 지내던 고혈압이 없었으며, 2022년 7월 29일부터 포나티닙 45 mg 복용을 시작하였다. 포나티닙 치료 시작 전 수축기 혈압(systolic blood pressure, sBP) 130-160 mmHg, 이완기 혈압(diastolic blood pressure, dBP) 70-90 mmHg 정도로 확인되었으나, CML-BP와 관련될 것으로 추정되는 38도 이상의 열이 동반되는 상태여서 혈압에 대해서는 보존적 치료를 하였다. 포나티닙 치료를 시작하고 열은 감소하였으나 경구 항고혈압제 복용에도 혈압은 계속 높게 측정되었고, 2023년 8월 3일 아침 혈압 176/98로 확인되어, 니페디핀(nifedipine) 정맥 지속 주입을 시작하였다. 약물 투여에도 혈압은 sBP 160-180 mmHg, dBP 80-100 mmHg 및 두통, 전신 통증 호소하여, 2023년 8월 11일에는 포나티팁 30 mg으로 감량하였다. 이후 혈압은 sBP 130-140 mmHg, dBP 70-80 mmHg가 유지되고, 증상 역시 호전되어, 정맥 항고혈압제를 중단하고 경구 항고혈압제로 변경하였다. 2022년 8월 24일 다시 포나티닙 45 mg으로 증량하였고, 1개월 골수검사에서 미성숙 세포 3%로 완전 반응(complete response, CR)으로 확인되었으며, 골수검사상 BCR-ABLIS 13.2088%로 확인되었다. 포나티닙 45 mg과 항고혈압제를 복용하며 특별히 호소하는 증상 없이 2022년 11월 18일 동종 조혈모세포이식을 시행하였다. 이식 후 3개월 검사 시 CCyR 반응 획득 및 BCR-ABLIS은 검출되지 않음으로 확인되었다.
CVE와 더불어 고혈압은 포나티닙을 복용하는 환자들에게 흔히 동반되는 부작용 중 하나이다. CML에 사용하는 다른 TKI들과는 달리 포나티닙이 ABL1 외에도 VEGFR2에도 결합하여 VEGF 신호 전달을 강하게 차단하는 것과 관련이 있을 것으로 추정되는데[13], VEGF 신호 전달은 신생 혈관 생성뿐만 아니라, 산화 질소(nitric oxide)와 프로스타글란딘(prostaglandin)과 같은 혈관 확장 물질을 증가시키는 역할을 하기 때문이다[13,22]. 쥐 모델 연구에서 포나티닙이 노르에피네프린(norepinephrine)에 의한 혈관 수축과 아세틸콜린(acetylcholine)에 의한 혈관 확장을 억제하며, 이러한 효과는 산화 질소와 프로스타글란딘, 그리고 칼슘 채널 차단제(calcium channel blocker)에 의해 상쇄되는 것으로 나타나서, 포나티닙의 고혈압 및 혈관 관련 합병증 기전을 뒷받침한다[26].
PACE 연구에서 3단계 이상의 중증 고혈압은 12%로 보고되었으나, 포나티닙 시작 전 고혈압 유무에 상관없이 68% 정도의 환자들이 혈압 상승을 경험한 것으로 나타났다(Table 2) [18]. 이에 비해 닐로티닙의 경우는 ENESTnd 5년 추적 결과에서 3단계 이상 고혈압은 1.1-1.4%, 이마티닙은 0.4%로 보고되었다[8]. 포나티닙의 용량을 조절한 OPTIC 연구에서는 전반적으로 약물 관련 부작용의 비율을 감소하였는데, 전체 28% 환자들이 고혈압을 보여주었으나, 그 중 대다수는 1,2단계 정도의 경증이었던 것으로 확인되었다(Table 2) [19]. 포나티닙의 혈관 관련 독성이 잘 알려짐에 따라 실제 진료 현장에서는 고혈압, CVE 등과 같은 혈관 관련 독성의 빈도를 오히려 더 낮게 보고하기도 한다. 단일 기관에서 포나티닙을 사용한 78명을 분석한 결과에 따르면, 대부분의 환자들이 45 mg으로 치료를 시작하였던 2014년 이전에는 심각한 CVE가 13.5%였던 반면, 혈관 관련 위험 요인에 따라 용량을 낮추어 시작한 환자들이 많았던 2014년 이후에는 CVE의 발생이 2.4% 정도로 줄었으며, 전체적으로 7.7% 정도의 환자만이 새로운 고혈압을 경험하였다고 보고하였다[27]. 해당 연구에서 흥미로웠던 점은 2014년 이후 순환기내과로 환자 협진 횟수는 오히려 의미 있게 늘어났다는 것인데, 부작용 감소를 위한 다학제적인 접근의 필요하다는 것을 시사한다.
CML 환자에서 TKI 치료와 관련된 혈관 관련 합병증의 높은 빈도를 생각한다면 CML 진단 시부터 혈관 관련 위험인자에 대한 충분한 조사가 필요하다(Table 3). 고혈압, 당뇨, 고지혈증과 같은 현재 병력, 이전 심혈관 관련 질환 여부 등의 과거력, 그리고 음주, 흡연 여부 등의 생활에 습관에 대한 문진이 필요하다. 환자가 닐로티닙이나 포나티닙 투약을 준비 중이라면 기본 혈액검사 외에도 당뇨, 고지혈증에 대한 혈액검사가 필요하며, 심전도와 심장 초음파, 그리고 발목 상완 지수(ankle-brachial index, ABI) 검사가 권장된다[13]. 특히 ABI는 비침습적 방법으로 혈관의 동맥경화 정도를 높은 민감도와 특이도로 확인할 수 있는 검사이므로 TKI 사용 전 환자의 혈관 관련 위험도를 예측하는 데 도움이 된다[28]. 환자가 혈관 관련 독성이 높을 것으로 예상된다면 포나티닙과 시작 전에는 스트레스 테스트, 심장 컴퓨터단층검사(cardiac computed tomography), 말초 혈관 초음파검사 등을 추가적으로 해 볼 수도 있다[29]. 치료 시작 전 Framingham risk score나, 유럽심장학회에서 제안한 Systematic Coronary Risk Evaluation Project (SCORE) 시스템을 이용하여 TKI 사용 전 심혈관 질환의 위험도를 체계적으로 예측할 수도 있다[9,30], SCORE 시스템 같은 경우는 닐로티닙 사용 환자에서 혈관 관련 부작용 예측에 의미가 있는 것으로 검증된 바 있으며[31], 포나티닙 사용 환자에게도 적용되어 CVE 발생을 예측하는 유일한 인자로 확인되기도 하였다[32]. 또한 진단 후 첫 번째 TKI 선택 시 놓치지 말아야 할 점은 환자의 기저 질환 및 TKI의 개별 부작용을 충분히 고려하여 치료 약제를 선택하는 것이다[33]. 가령 이전 중풍, 심근경색의 이력이 있거나, 당뇨, 고혈압이 있다면 닐로티닙 대신 다사티닙, 이마티닙 같은 TKI를 사용하는 것이 권장된다.
심혈관 질환의 위험을 낮추기 위해 일반인들에게 권장되었던 ABCDE 스텝을 CML 환자에게도 적용해 볼 수 있다 (Figure 2) [13,28,33]. 즉, 환자에게 심혈관 질환의 증상에 대해 교육하고, 예방적으로 아스피린을 투약하며, 당뇨 또는 고지혈증 등과 같이 동맥경화를 악화 시키는 요인들이 있다면 적극적으로 약물 치료를 하고, 식사, 체중 감소, 운동 등 라이프스타일 교정 등을 실시하여 CVE 발생을 줄이는 노력을 하는 것이다. 그러나 아스피린과 같은 약제를 예방적으로 사용하는 것에는 다소 논란이 있을 수 있다. 이는 저 용량 아스피린을 계속 투여 중이었음에도 불구하고 닐로티닙 투약을 받은 환자의 혈액검사에서 혈소판 부착 능력의 증가와 함께, 염증, 혈전 생성 관련 단백질의 높게 측정되며[12], 포나티닙은 오히려 혈소판의 기능을 억제한다는 연구결과도 존재하기 때문이다[21]. 그러나 실제 임상 경험에서 예방적 아스피린의 투약이 효과가 있었다는 연구도 있어서 이득을 보는 특정 환자군이 있을 것으로 생각 된다[32,34]. 향후 예방적 항혈소판제의 투약에 대한 전향적 연구가 요구된다.
심혈관 질환의 위험을 낮추기 위해 일반인들에게 권장되었던 ABCDE 스텝을 CML 환자에게도 적용해 볼 수 있다(Figure 2) [13,28,33]. 즉, 환자에게 심혈관 질환의 증상에 대해 교육하고, 예방적으로 아스피린을 투약하며, 당뇨 또는 고지혈증 등과 같이 동맥경화를 악화시키는 요인들이 있다면 적극적으로 약물 치료를 하고, 식사, 체중 감소, 운동 등 라이프스타일 교정 등을 실시하여 CVE 발생을 줄이는 노력을 하는 것이다. 그러나 아스피린과 같은 약제를 예방적으로 사용하는 것에는 다소 논란이 있을 수 있다. 이는 저 용량 아스피린을 계속 투여 중이었음에도 불구하고 닐로티닙 투약을 받은 환자의 혈액검사에서 혈소판 부착 능력의 증가와 함께, 염증, 혈전 생성 관련 단백질의 높게 측정되며[12], 포나티닙은 오히려 혈소판의 기능을 억제한다는 연구결과도 존재하기 때문이다[21]. 그러나 실제 임상 경험에서 예방적 아스피린의 투약이 효과가 있었다는 연구도 있어서 이득을 보는 특정 환자군이 있을 것으로 생각된다[32,34]. 향후 예방적 항혈소판제의 투약에 대한 전향적 연구가 요구된다.
치료 중 환자의 혈관 관련 위험인자 발생을 주기적으로 관찰하고 닐로티닙이나 포나티닙과 같은 약물을 복용 중이라면, 3-6개월 간격으로 당뇨, 고지혈증 검사, 심전도와 심장 초음파, 그리고 ABI 검사를 주기적으로 시행하는 것이 권장된다(Table 2) [13]. 포나티닙을 사용하는 환자에서 고혈압은 약제 투약을 시작하고 며칠 이내에 발생할 가능성이 높으므로, 초기부터 주기적인 혈압 측정이 권장된다[13]. TKI 사용 시 고혈압 부작용은 VEGF의 억제에 의한 혈관 수축에 의한 것임을 고려한다면, 디히드로피리딘(dihydropyridine) 계열의 칼슘 채널 억제제가 도움이 될 수 있다. 그러나 비디히드로피리딘 계열의 칼슘 채널 억제제는 CYP3A4 억제 기능이 있어 TKI와 약물 상호 작용을 일으킬 수 있으므로 사용에 주의가 필요하다[35]. 그리고 치료 반응이 충분히 있다면 적극적으로 약물의 농도를 조절하는 전략이 필요하다. 만약 환자가 심혈관 관련 증상을 호소하거나, 혈관 관련한 위험인자들이 새로이 확인된다면 관련 과로 적극적인 협진을 의뢰하는 등 다제학적 접근을 고려해야 하겠다.
혈관 관련 위험인자가 있고 오랜 기간 TKI 사용으로 CVE 부작용이 우려되는 경우, 기능적 완치(treatment-free remission, TFR)를 목표로 치료 중단도 고려해 볼 수 있다. TFR을 위한 연구 프로토콜이나 가이드라인에 따라 기준은 다소 차이가 있으나, TFR 시도를 위해서는 적어도 5년 이상의 TKI 치료기간과 적어도 2-3년 이상의 깊은 분자학적 반응(deep molecular response, DMR) 유지기간이 필요하다[20]. ENESTnd 연구에서 DMR보다 좀 더 깊은 반응인 완전 반응(molecular response 4.5, MR 4.5)을 기준으로 TFR 시도가 가능한 환자군을 추정하였을 때, 10년 정도 투약하면 이마티닙은 29.7%가 가능한 반면, 닐로티닙 300 mg 1일 2회 복용한 환자들은 48.6%가 TFR 시도가 가능한 것으로 나타났다[10]. 따라서 CVE 위험성이 높은 닐로티닙과 같은 약제 투약시에는 적극적인 TFR 시도가 혈관 관련 합병증 발생을 예방하는 방법이 될 수 있다.
TKI의 도입은 CML 환자의 기대 여명을 일반인과 유사한 수준으로 끌어올렸을 뿐만 아니라[36], 경구 제재라는 매우 큰 편리함을 환자에게 안겨주었다. 그러나 대부분의 TKI 약제들이 그러하듯, CML에 사용하는 TKI들도 치료 표적과 동시에 다른 인산화효소에도 영향을 끼치는 오프 타켓(off-target) 효과를 가지고 있다[13]. 따라서 CML 환자의 TKI 관련한 독성의 상당수는 여기에서 기인할 것으로 추정된다. 치료 경험과 연구의 누적으로 치료하고자 하는 표적에만 작용하여 부작용을 최소한으로 낮출 수 있는 TKI의 개발에 대한 노력은 계속되고 있으나, 아직까지 부작용에서 완전히 자유로울 수는 없다. 따라서 환자와 의료진 모두 치료 약제에 대한 충분한 지식을 가지고 부작용에 대한 관찰이 요구된다. 특히 혈관 관련 독성과 같이 환자의 생명을 위태롭게 할 수 있는 부작용에 대해서는 아무리 큰 관심을 보이더라도 지나치지 않으므로, 면밀한 증상의 관찰과 충분한 검사를 통해 위험한 환자들을 조기에 분류하고, 막을 수 있는 부작용은 놓치지 않는 노력이 그 어느 때보다 필요하다.
Figure 1.
The mechanisms of cardiovascular events occurring owing to nilotinib. Illustrated by the author.

Figure 2.
ABCDE steps to reduce cardiovascular events. CVE, cardiovascular event. Illustrated by the author.
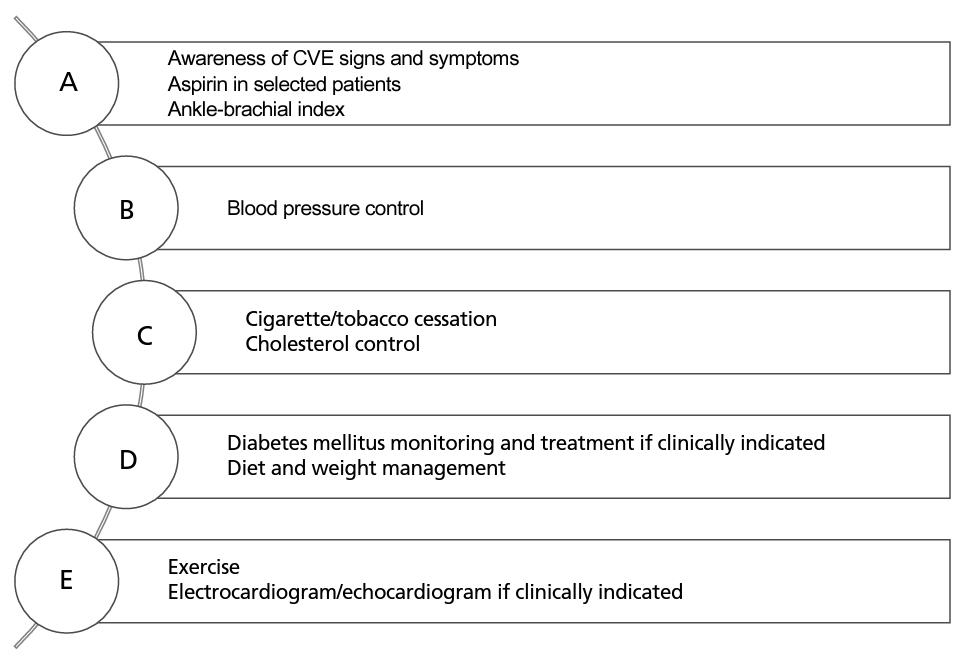
Table 1.
Cardiovascular events in frontline randomized trial of nilotinib
| Framingham risk of CVEs |
ENESTnd |
||
|---|---|---|---|
| Nilotinib 300 mga) | Nilotinib 400 mga) | Imatinib 400 mgb) | |
| All CVEs | |||
| CVEs occurrence before 5 yr | |||
| <10% | 2.2 (4/178) | 4.0 (7/176) | 0.5 (1/182) |
| ≥10% to <20% | 12.2 (5/41) | 19.2 (10/52) | 4.1 (2/49) |
| ≥20% | 15.0 (6/40) | 28.9 (11/38) | 3.0 (1/33) |
| CVEs occurrence after 5 yr | |||
| <10% | 8.7 (10/115) | 15.2 (19/125) | 1.1 (1/94) |
| ≥10% to <20% | 28.0 (7/25) | 48.5 (16/33) | 17.4 (4/23) |
| ≥20% | 33.3 (8/24) | 33.3 (7/21) | 4.8 (1/21) |
Values are presented as % (number/total number).
Modified from Kantarjian HM, et al. Leukemia 2021;35:440-453, according to the Creative Commons license [10].
CVE, cardiovascular event.
Table 2.
Cardiovascular events and hypertension in clinical trials of ponatinib and asciminib
|
Ponatinib |
Asciminib |
|||||
|---|---|---|---|---|---|---|
|
PACE [18] |
OPTIC [19] |
STAMP [25] |
||||
| Ponatinib 45 mga) | Ponatinib 45 mga) | Ponatinib 30 mga) | Ponatinb 15 mga) | Asciminib 40 mgb) | Bosutinib 500 mga) | |
| All CVEs | 31.1 (84/270)c) | 9.6 (9/94) | 5.3 (5/94) | 3.2 (3/94) | 5.1 (8/156) | 1.3 (1/76) |
| HTN | 36.7 (99/270)c) | 27.7 (26/94) | 26.6 (25/94) | 23.4 (22/94) | 13.5 (21/156) | 5.3 (4/76) |
Values are presented as % (number/total number).
Based on Cortes JE, et al. Blood 2018;132:393-404, Cortes J, et al. Blood 2021;138:2042-2050, and Hochhaus A, et al. Leukemia 2023;37:617-626, according to the Creative Commons license [18,19,25]
PACE, Ponatinib Ph+ ALL and CML Evaluation; OPTIC, Optimizing Ponatinib Treatment in CP-CML; STAMP, Specifically Targets the ABL Myristoyl Pocket; CVE, cardiovascular event; HTN, hypertension; CML-CP, chronic myeloid leukemia-chronic phase.
Table 3.
Recommendations for cardiovascular risk assessment
References
3. O’Brien SG, Guilhot F, Larson RA, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 2003;348:994-1004.


4. Shoukier M, Kubiak M, Cortes J. Review of new-generation tyrosine kinase inhibitors for chronic myeloid leukemia. Curr Oncol Rep 2021;23:91.


5. Saglio G, Kim DW, Issaragrisil S, et al. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med 2010;362:2251-2259.


6. Le Coutre P, Rea D, Abruzzese E, et al. Severe peripheral arterial disease during nilotinib therapy. J Natl Cancer Inst 2011;103:1347-1348.


7. Aichberger KJ, Herndlhofer S, Schernthaner GH, et al. Progressive peripheral arterial occlusive disease and other vascular events during nilotinib therapy in CML. Am J Hematol 2011;86:533-539.


8. Hochhaus A, Saglio G, Hughes TP, et al. Long-term benefits and risks of frontline nilotinib vs imatinib for chronic myeloid leukemia in chronic phase: 5-year update of the randomized ENESTnd trial. Leukemia 2016;30:1044-1054.



9. D’Agostino RB Sr, Vasan RS, Pencina MJ, et al. General cardiovascular risk profile for use in primary care: the Framingham Heart Study. Circulation 2008;117:743-753.


10. Kantarjian HM, Hughes TP, Larson RA, et al. Long-term outcomes with frontline nilotinib versus imatinib in newly diagnosed chronic myeloid leukemia in chronic phase: ENESTnd 10-year analysis. Leukemia 2021;35:440-453.



11. Hadzijusufovic E, Albrecht-Schgoer K, Huber K, et al. Nilotinib-induced vasculopathy: identification of vascular endothelial cells as a primar y target site. Leukemia 2017;31:2388-2397.



12. Alhawiti N, Burbury KL, Kwa FA, et al. The tyrosine kinase inhibitor, nilotinib potentiates a prothrombotic state. Thromb Res 2016;145:54-64.


13. Moslehi JJ, Deininger M. Tyrosine kinase inhibitor-associated cardiovascular toxicity in chronic myeloid leukemia. J Clin Oncol 2015;33:4210-4218.



14. Lassila M, Allen TJ, Cao Z, et al. Imatinib attenuates diabetes-associated atherosclerosis. Arterioscler Thromb Vasc Biol 2004;24:935-942.


15. Fitter S, Vandyke K, Schultz CG, White D, Hughes TP, Zannettino AC. Plasma adiponectin levels are markedly elevated in imatinib-treated chronic myeloid leukemia (CML) patients: a mechanism for improved insulin sensitivity in type 2 diabetic CML patients? J Clin Endocrinol Metab 2010;95:3763-3767.


16. Giles FJ, Mauro MJ, Hong F, et al. Rates of peripheral arterial occlusive disease in patients with chronic myeloid leukemia in the chronic phase treated with imatinib, nilotinib, or non-tyrosine kinase therapy: a retrospective cohort analysis. Leukemia 2013;27:1310-1315.


17. Groarke JD, Cheng S, Moslehi J. Cancer-drug discovery and cardiovascular surveillance. N Engl J Med 2013;369:1779-1781.


18. Cortes JE, Kim DW, Pinilla-Ibarz J, et al. Ponatinib efficacy and safety in Philadelphia chromosome-positive leukemia: final 5-year results of the phase 2 PACE trial. Blood 2018;132:393-404.



19. Cortes J, Apperley J, Lomaia E, et al. Ponatinib dose-ranging study in chronic-phase chronic myeloid leukemia: a randomized, open-label phase 2 clinical trial. Blood 2021;138:2042-2050.



20. Hochhaus A, Baccarani M, Silver RT, et al. European LeukemiaNet 2020 recommendations for treating chronic myeloid leukemia. Leukemia 2020;34:966-984.



21. Loren CP, Aslan JE, Rigg RA, et al. The BCR-ABL inhibitor ponatinib inhibits platelet immunoreceptor tyrosine-based activation motif (ITAM) signaling, platelet activation and aggregate formation under shear. Thromb Res 2015;135:155-160.



22. Bair SM, Choueiri TK, Moslehi J. Cardiovascular complications associated with novel angiogenesis inhibitors: emerging evidence and evolving perspectives. Trends Cardiovasc Med 2013;23:104-113.



23. Hughes TP, Mauro MJ, Cortes JE, et al. Asciminib in chronic myeloid leukemia after ABL kinase inhibitor failure. N Engl J Med 2019;381:2315-2326.



24. Réa D, Mauro MJ, Boquimpani C, et al. A phase 3, open-label, randomized study of asciminib, a STAMP inhibitor, vs bosutinib in CML after 2 or more prior TKIs. Blood 2021;138:2031-2041.



25. Hochhaus A, Réa D, Boquimpani C, et al. Asciminib vs bosutinib in chronic-phase chronic myeloid leukemia previously treated with at least two tyrosine kinase inhibitors: longer-term follow-up of ASCEMBL. Leukemia 2023;37:617-626.



26. Hadzijusufovic E, Kirchmair R, Theurl M, et al. Ponatinib exerts multiple effects on vascular endothelial cells: possible mechanisms and explanations for the adverse vascular events seen in CML patients treated with ponatinib. Blood 2016;128:1883.

27. Chan O, Talati C, Isenalumhe L, et al. Side-effects profile and outcomes of ponatinib in the treatment of chronic myeloid leukemia. Blood Adv 2020;4:530-538.



28. Manouchehri A, Kanu E, Mauro MJ, Aday AW, Lindner JR, Moslehi J. Tyrosine kinase inhibitors in leukemia and cardiovascular events: from mechanism to patient care. Arterioscler Thromb Vasc Biol 2020;40:301-308.

29. Casavecchia G, Galderisi M, Novo G, et al. Early diagnosis, clinical management, and follow-up of cardiovascular events with ponatinib. Heart Fail Rev 2020;25:447-456.


30. Perk J, De Backer G, Gohlke H, et al. European guidelines on cardiovascular disease prevention in clinical practice (version 2012) : the fifth joint task force of the European society of cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of nine societies and by invited experts). Int J Behav Med 2012;19:403-488.

31. Breccia M, Molica M, Zacheo I, Serrao A, Alimena G. Application of systematic coronary risk evaluation chart to identify chronic myeloid leukemia patients at risk of cardiovascular diseases during nilotinib treatment. Ann Hematol 2015;94:393-397.


32. Caocci G, Mulas O, Abruzzese E, et al. Arterial occlusive events in chronic myeloid leukemia patients treated with ponatinib in the real-life practice are predicted by the Systematic Coronary Risk Evaluation (SCORE) chart. Hematol Oncol 2019;37:296-302.



34. Caocci G, Mulas O, Annunziata M, et al. Cardiovascular toxicity in patients with chronic myeloid leukemia treated with second-generation tyrosine kinase inhibitors in the real-life practice: identification of risk factors and the role of prophylaxis. Am J Hematol 2018;93:E159-E161.


Peer Reviewers’ Commentary
만성골수성백혈병은 계속되는 타이로신인산화효소(tyrosine kinase) 활성도를 나타내는 유전자에 의해 발생하는 골수증식 종양이다. 타이로신인산화효소억제제(tyrosine kinase inhibitor, TKI)는 만성골수성백혈병 환자의 치료 결과를 극적으로 향상시켰으나, 약제를 장기간 복용해야 하므로 약제의 독성, 특히 심혈관계 독성이 문제가 된다. 이 논문은 두 증례 보고와 함께 심혈관계 독성의 역학과 병태생리에 대한 고찰과 함께 고혈압과 심혈관계 합병증 발생을 예방하기 위한 위험인자 관리와 모니터링 방법에 관해 무작위배정연구 결과를 광범위하게 분석하여 정리하였다. TKI 유발 혈관 독성의 전체적인 기전은 총체적으로 밝혀지지는 않았으나 여러 요소가 개입된다고 알려져 있다. TKI를 사용해 치료할 때 심혈관계 부작용을 예방하고 감소시키는 치료 전략은 만성골수성백혈병 환자의 치료에서 무엇보다 중요하다. 이 논문은 TKI로 만성골수성백혈병 환자를 치료하는 임상 현장에 좋은 지침이 될 것으로 판단된다.
[정리: 편집위원회]
-
METRICS

-
- 1 Crossref
- Scopus
- 1,662 View
- 97 Download
-
Related articles in
J Korean Med Assoc






